
Xpert® Xpress MVP
Multiplex vaginal panel that enables results within 60 minutes from a single specimen for Bacterial Vaginosis, Vulvovaginal Candidiasis, and Trichomoniasis
Sign in or create a MyCepheid account to add items to cart
Test pack size(s)

10 Tests
XPRSMVP-10
Qty
Unit price
Subtotal
USD
Product is not available for purchase in your region.
Collection devices
Swab Specimen Collection Kit (Pack of 50)
SWAB/G-50-US
Qty
Unit price
Subtotal
USD
Product is not available for purchase in your region.
Total
{{currency}}
0
Error adding items to cart. If this error persists, please contact Digital Support
The Need
Vaginitis is a leading cause of clinic visits by women, with more than 10 million per year in the United States.1 Limitations with traditional test methods contribute to poor diagnosis, inappropriate treatment, and persistent symptoms.2
High performing PCR tests are available for large, batch analyzers but have limited practical use for clinicians due to a lag in reportable results, making appropriate same day treatment a challenge.3
Challenges remain:
High performing PCR tests are available for large, batch analyzers but have limited practical use for clinicians due to a lag in reportable results, making appropriate same day treatment a challenge.3
Challenges remain:
- Detection and differentiation of organisms associated with bacterial vaginosis (BV), vulvovaginal candidiasis (VVC), and trichomoniasis (TV), which all present with similar symptoms
- Mixed and co-infections of BV, VVC, and TV are common, further complicating diagnosis and the guidance of appropriate treatment4,5
- Testing options that offer accurate and objective test results to drive timely and appropriate treatment for improved patient management
1 Brown H, et al. Improving the Diagnosis of Vulvovaginitis: Perspectives to Align Practice, Guidelines, and Awareness. Population Health Management 2020 23:S1, S-3-S-12
2 Hillier SL, et al. Diagnosis and treatment of vaginal discharge syndromes in community practice settings. Clin Infect Dis. 2021 May 04; 72(9) 1538-1543
3 Gaydos CA, et al. Use of a Rapid Diagnostic for Chlamydia trachomatis and Neisseria gonorrhoeae for Women in the Emergency Department Can Improve Clinical Management: Report of a Randomized Clinical Trial. Ann Emerg Med. 2019 Jul; 74(1) 36-44
4 Gaydos CA, et al. Clinical Validation of a Test for the Diagnosis of Vaginitis. Obstetrics & Gynecology: July 2017 - Volume 130 - Issue 1 - p 181-189 doi: 10.1097/ AOG.0000000000002090
5 Schwebke J, et al. Clinical Validation of the Aptima Bacterial Vaginosis and Aptima Candida/Trichomonas Vaginitis Assays: Results from a Prospective Multicenter Clinical Study. J Clin Microbiol 58:e01643-19.
2 Hillier SL, et al. Diagnosis and treatment of vaginal discharge syndromes in community practice settings. Clin Infect Dis. 2021 May 04; 72(9) 1538-1543
3 Gaydos CA, et al. Use of a Rapid Diagnostic for Chlamydia trachomatis and Neisseria gonorrhoeae for Women in the Emergency Department Can Improve Clinical Management: Report of a Randomized Clinical Trial. Ann Emerg Med. 2019 Jul; 74(1) 36-44
4 Gaydos CA, et al. Clinical Validation of a Test for the Diagnosis of Vaginitis. Obstetrics & Gynecology: July 2017 - Volume 130 - Issue 1 - p 181-189 doi: 10.1097/ AOG.0000000000002090
5 Schwebke J, et al. Clinical Validation of the Aptima Bacterial Vaginosis and Aptima Candida/Trichomonas Vaginitis Assays: Results from a Prospective Multicenter Clinical Study. J Clin Microbiol 58:e01643-19.
The Solution
The Xpert® Xpress MVP test is a CLIA-waived FDA cleared on-demand PCR test to aid in the diagnosis of vaginal infections in symptomatic women within an hour. This test introduces a simpler algorithm for organisms associated with bacterial vaginosis, combined with the detection of Candida species associated with vulvovaginal candidiasis, and Trichomonas vaginalis, from a single sample on Cepheid’s easy-to-use GeneXpert® systems.
On-demand molecular testing — an ideal solution:
On-demand molecular testing — an ideal solution:
- Actionable diagnostic aid for bacterial vaginosis, vulvovaginal candidiasis, and trichomoniasis within 60 minutes, including potential co-infections
- On-demand results for 4 separate callouts, bacterial vaginosis, Candida group,* C. glabrata/C. krusei, and T. vaginalis from a single sample collection
- Implementing easy-to-use testing with GeneXpert technology
- Approved for women 14 years old and over
The Impact
- The Xpert Xpress MVP demonstrates high sensitivity and specificity in detecting the most common causes of vaginitis from clinician and patient-collected samples in both laboratory-based and point-of-care settings.6
- Xpert Xpress MVP is a flexible, easy to use, on-demand test designed with optimized workflow efficiencies for the diagnosis of vaginitis-like symptoms.
- Accurate differential diagnosis with Xpert Xpress MVP informs appropriate treatment on time, first time, potentially improving clinical outcomes.
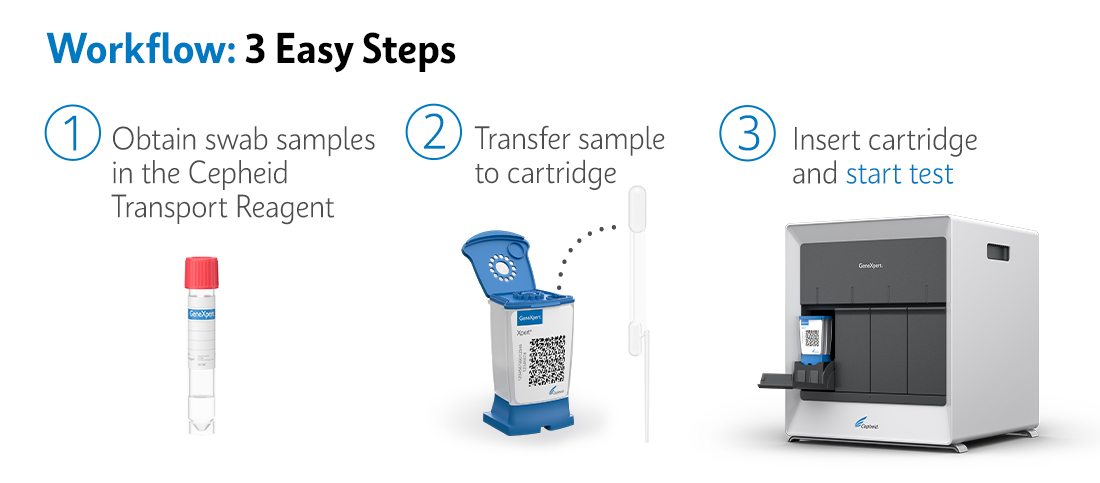
Workflow: 3 Easy Steps using the GeneXpert® System
6 Lillis, RA, et al. Clinical Evaluation of a New Molecular Test for the Detection of Organisms Causing Vaginitis and Vaginosis. J Clin Microbiol. 2023 Mar 23;61(3):e0174822. doi: 10.1128/jcm.01748-22. Epub 2023 Feb 28.
* Detected organisms not reported individually.
^ Target includes C. albicans, C. tropicalis, C. parapsilosis, and C. dubliniensis
# Contrived specimens were prepared using individual negative clinical vaginal swab specimens.
Refer to most current Instructions for Use 301-8994 and 302-6886 (for GeneXpert Dx and Infinity Systems) for complete details
US-IVD. In Vitro Diagnostic Medical Device.
* Detected organisms not reported individually.
^ Target includes C. albicans, C. tropicalis, C. parapsilosis, and C. dubliniensis
# Contrived specimens were prepared using individual negative clinical vaginal swab specimens.
Refer to most current Instructions for Use 301-8994 and 302-6886 (for GeneXpert Dx and Infinity Systems) for complete details
US-IVD. In Vitro Diagnostic Medical Device.





